INTRODUCTION
In our June 2025 newsletter to physicians, Lenire® was highlighted as a tinnitus treatment option. This included positive real-world outcomes from a retrospective review at the Alaska Hearing & Tinnitus Center, reinforcing that bimodal neuromodulation can deliver meaningful benefits for patients with moderate-to-severe tinnitus. Sufficient internal data is now available to assess early outcome trends within our own patient population. The following analysis summarizes these clinic results.
METHODS
The Tinnitus Handicap Inventory (THI), a validated 25-item questionnaire widely used to assess tinnitus-related handicap [2], was utilized to monitor patient progress throughout treatment. A total of 18 individuals were included in the analysis. Of these, five were excluded from the current review: three had not yet reached the 12-week treatment mark, and two discontinued treatment for reasons unrelated to clinical outcomes. The mean patient age was 66.8 years old. Of the 13 patients, 8 had a documented hearing loss, and 7 were actively using prescription hearing aids.
A THI was completed prior to treatment, at 6 weeks, 12 weeks, and 20+ weeks, when available. A clinically significant response was defined as >7-point improvement (reduction) in THI scores, reflecting established MCID criteria [3], consistent with published Lenire® studies.
RESULTS
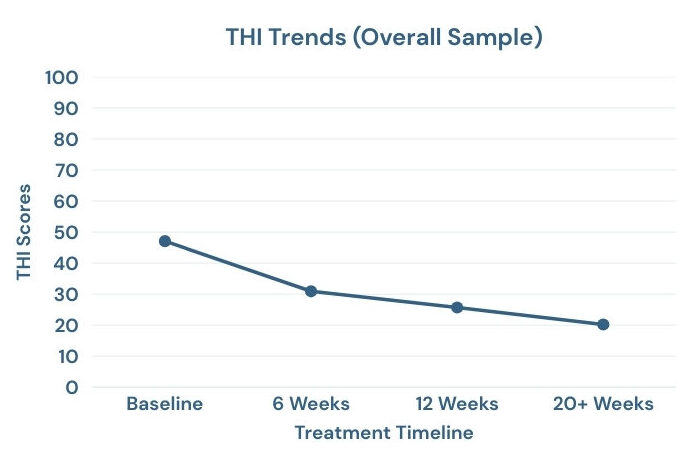
In the overall sample, the average total reduction in THI score was 28.31 points. At 12 weeks, 92% of patients achieved clinically significant improvement. We observed a progressive decline in THI scores across each time interval, with continued improvement in patients who reached 20+ weeks.
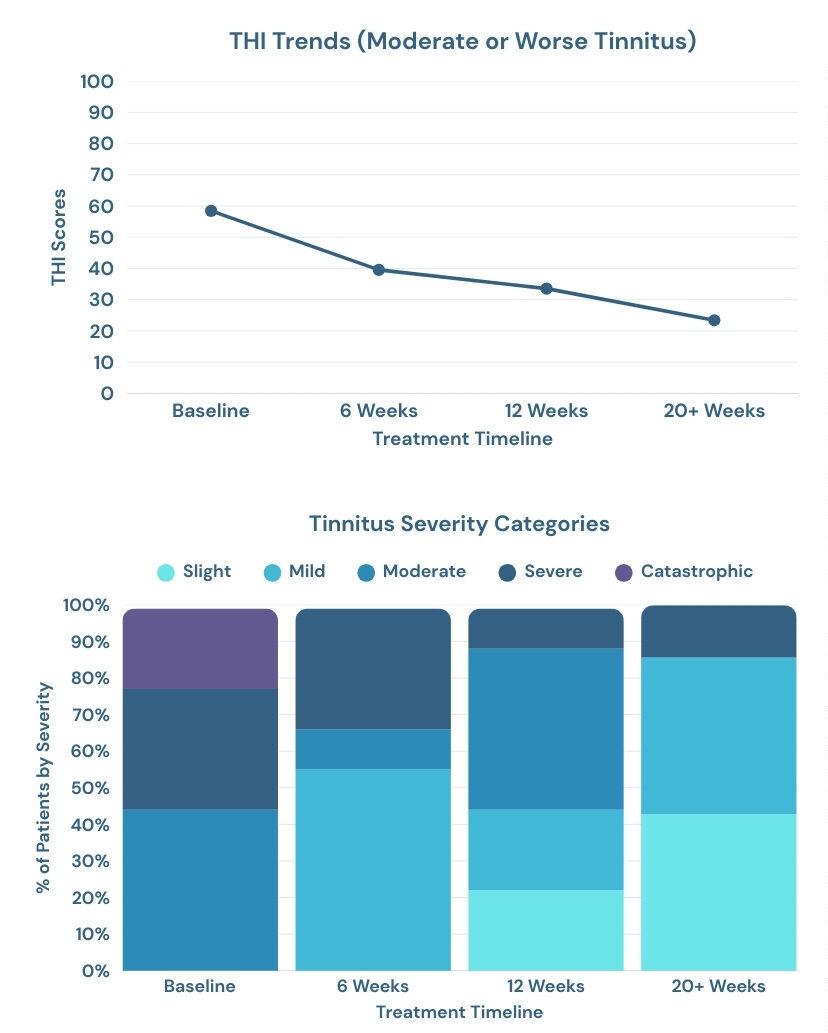
When greater baseline severity was reported, a stronger response pattern was observed. For individuals whose tinnitus was classified as ‘moderate’ or worse (THI >38), 100% of patients demonstrated a clinically significant improvement in their tinnitus at 12 weeks of treatment, with a 35.01 average reduction in THI scores.
When examining changes in tinnitus severity categories over time, a clear shift toward lower perceived handicap was observed. At baseline, within the subgroup reporting moderate tinnitus or greater (THI > 38), 22% were classified as catastrophic (78–100; severe disruption to daily life), 33% as severe (58–76; significant functional impact), and 44% as moderate (38–56; noticeable and bothersome in daily situations).
By the 12 week mark, the distribution shifted substantially: 11% reported severe tinnitus, 44% reporting moderate tinnitus, 22% reported mild (18–36; generally manageable), and 22% reported slight (0–16; minimal handicap). Improvement continued through the 20+ week mark among those who progressed to that stage of treatment.
Categorically, three individuals improved by one severity level, three improved by two levels, and three improved by three levels, reinforcing that the reduction in THI scores translated into meaningful improvements in functional severity classification.
CLINICAL CONTEXT
The THI remains one of the most widely used validated outcome measures for tinnitus impact and treatment response [5]. Importantly, emerging data suggests that MCID thresholds may vary depending on baseline severity and time interval, with some analyses estimating a 12-week MCID for THI closer to 11 points [2]. In our sample, the average improvement of 28.31 points substantially exceeds both traditional and more conservative MCID thresholds and was accompanied by meaningful reductions in severity classification, particularly among those presenting with moderate or more severe tinnitus at baseline.
SUMMARY
Although our sample size is small, our internal findings are consistent with larger-scale studies evaluating the efficacy of Lenire®. A steady reduction in THI scores was observed over time, with a stronger response pattern among individuals presenting with moderate or greater tinnitus severity at baseline. The majority of patients demonstrated clinically meaningful improvement within 12 weeks of initiating treatment. Outcomes will continue to be monitored as additional patients complete the protocol. Please consider referring your patients who are experiencing persistent, bothersome tinnitus for a comprehensive tinnitus evaluation to assess candidacy and review appropriate treatment options.
Sources:
[1] McMahan EE, Lim HH. Retrospective chart review demonstrating effectiveness of bimodal neuromodulation for tinnitus treatment in a clinical setting. Commun Med (Lond). 2025 Apr 28;5(1):112. doi: 10.1038/s43856-025-00837-3. PMID: 40295853; PMCID: PMC12037789.
[2] Engelke, M., et al P. (2025). Estimation of Minimal Clinically Important Difference for Tinnitus Handicap Inventory and Tinnitus Functional Index. Otolaryngology–head and neck surgery : official journal of American Academy of Otolaryngology-Head and Neck Surgery, 173(1), 69–79. https://doi.org/10.1002/ohn.1217
[3] Zeman, F., et al. (2011). Tinnitus handicap inventory for evaluating treatment effects: which changes are clinically relevant?. Otolaryngology–head and neck surgery : official journal of American Academy of Otolaryngology-Head and Neck Surgery, 145(2), 282–287. https://doi.org/10.1177/0194599811403882
[4] Boedts, M., et al (2024). Combining sound with tongue stimulation for the treatment of tinnitus: a multi-site single-arm controlled pivotal trial. Nature communications, 15(1), 6806. https://doi.org/10.1038/s41467-024-50473-z
[5] Sherlock, L. P., Ballard-Hernandez, J., Boudin-George, A., Clark, K., Colandrea, M., Edmonds, C., Kelley, C., Lovelace, S., Mahmood, S., Martinez, I., Myers, P., Pulliam, S., Sall, J., Spencer, M., Theodoroff, S. M., Tolisano, A. M., Wayman, L. M., Zaugg, T., & Folmer, R. L. (2025). Clinical Practice Guideline for Management of Tinnitus: Recommendations From the US VA/DOD Clinical Practice Guideline Work Group. JAMA otolaryngology– head & neck surgery, 151(5), 513–520. https://doi.org/10.1001/jamaoto.2025.0052